
PrEP to be listed on the PBS and subsidised from the start of April
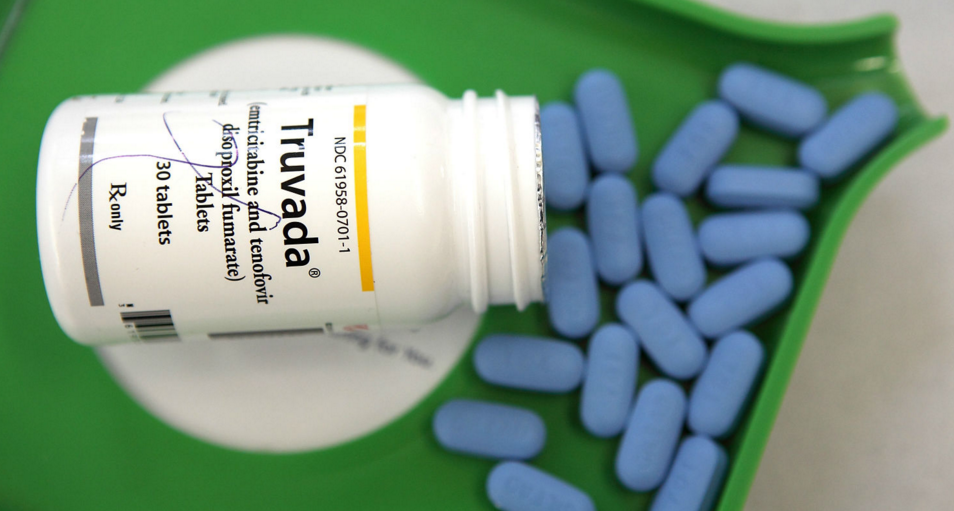
Pre-exposure prophylaxis (PrEP), the pill that prevents HIV, is about to be listed on the Pharmaceutical Benefits Scheme (PBS).
The $180 million subsidy will dramatically reduce the cost of the medication, known by the brand name Truvada.
The PBS allows Medicare card holders access to medications at reduced rates.
Federal Health Minister Greg Hunt announced today that PrEP will be listed on the scheme from April 1.
The $180 million listing means up to 32,000 patients each year will pay a maximum of only $39.50 per script, with concessional patients paying just $6.40.
Without subsidy patients would pay $2,496 per year for this medicine.
HIV experts initially raised concerns that doctors may not have the capacity for prescribing PrEP once it becomes available through the Pharmaceutical Benefits Scheme.
PrEP has until now been available to people at risk of HIV in some states through clinical trials, or by importing the expensive drug privately.
The medication is at least 99 per cent effective in preventing transmission of HIV when taken daily by HIV-negative people.
“For too long, people living with HIV have borne the brunt of expectation, responsibility, and blame when it comes to keeping the community safe from HIV,” said Nic Holas of HIV group The Institute of Many.
“PrEP offers HIV-negative people the opportunity to take more responsibility for their own safety.
“We hope this is the beginning of the end for new HIV transmissions in Australia, but we also know this is not the end of HIV stigma and discrimination.”









If you are HIV+ you do not pay anything for your meds
It’s about time. The NGO’s responsible for the delays need to be asked why it took so long to be listed on the PBS.
The Swiss, the CDC and FDA all recognised the benefit of Prep as much as 3 years ago !!
It will cost the PBS millions of dollars supporting those who have been infected, for the rest of their lives or until the drug cartel gets off it’s back side and speeds up the development of a vaccine and cure.
I guess we all know why they wont.
They are making more money selling their woeful ART and their nasty side effects which impact so many !.