
Australia on HIV home testing back foot
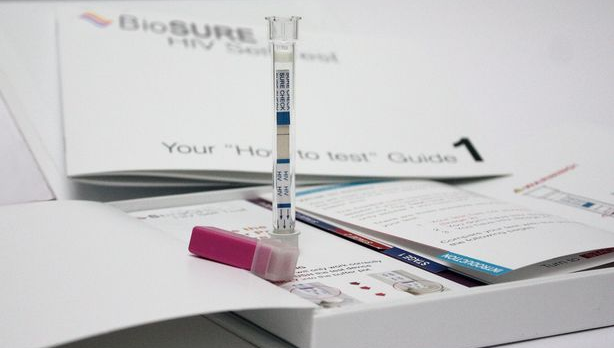
THE introduction of HIV home testing kits in the UK this week has further highlighted its lack of availability in Australia, LGBTI health bodies have said, hampering efforts to increase testing rates.
The sale of the Bio Sure kit in England, Wales and Scotland means both British and American consumers now have access to home testing which can give an indicative HIV result in just 15 minutes.
Last year the Therapeutic Goods Administration (TGA) lifted restrictions on the sale of home testing kits in Australia.
However, no manufacturer has applied for authorisation to sell the kits domestically.
Victorian AIDS Council (VAC) chief executive Simon Ruth said Australia had been too slow to give the green light to rapid testing, including home kits.
“The speed at which rapid HIV testing technology was introduced in Australia has been highly criticised,” he told the Star Observer.
“We know firsthand that making HIV testing convenient and accessible improves outcomes in HIV prevention and treatment.
“Australia needs to keep pace with countries like the US or the UK if we’re to reach the goals outlined in the seventh national HIV strategy.”
The strategy, which was launched last year, aims to reduce new HIV infections in Australia to zero by 2020.
“The approval of HIV home testing kits could reduce the time between HIV infection and diagnosis, thereby giving individuals the best chance at managing their ongoing health and wellbeing as well as reducing the possibility of onward transmission,” Ruth said.
While home testing kits are not yet available, they are similar to the kits used at the VAC’s PRONTO! and ACON’s a[TEST] rapid testing centres such which can provide results in as little as 30 minutes.
In February, ACON opened the country’s first permanent and dedicated HIV rapid testing centre on Sydney’s Oxford St.
ACON chief executive Nicolas Parkhill said home testing would be an “important addition” to testing options already available including GPs, sexual health clinics and community-based services such as a[TEST].
“What is needed now is for a manufacturer to put forth an application to the TGA and we will be encouraging them to do so.”
Talking to the Star Observer in December, Parkhill said the requirements for TGA approval “are probably too high and too expensive” for the kits.
However, he said new TGA guidelines — released last month— meant it was now easier for the kits to be signed off for sale.
Parkhill cautioned that if the kits were introduced they should include information on the need for confirmatory testing and support services.
“That way, if a person does test positive, appropriate support and treatment advice will be easily available,” he said.
Also in February, ACON launched a campaign to encourage men who have tested HIV positive to treat earlier to reduce transmission rates and potentially improve their own health.









Australia as per usual always behind the times on marriage equality, HIV testing reforms, transport and internet speeds!
What a total embarrassing joke this country – it really truly is!
I have had enough of this stupid Government, unprofessionally run by KKK Taliban Liberal party monster called Tony Abbott!