
Long-Acting HIV PrEP Drug May Be Available In 2022
The first long-acting HIV prevention medication may be available in the United States as early as the first quarter of 2022. This news comes even as Moderna announced last month that it was set to start human trials of its new HIV mRNA vaccines.
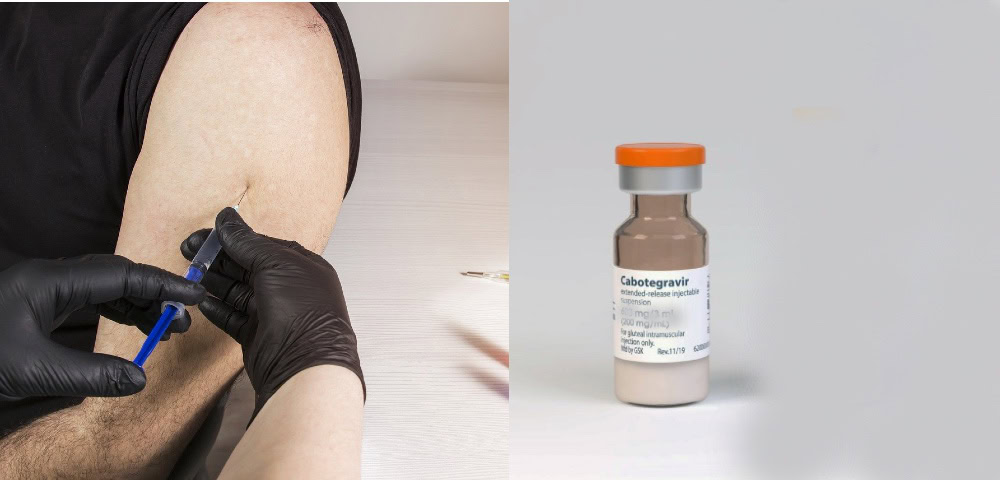
On September 23, the US Food and Drug Administration (FDA) granted priority review status for a New Drug Application (NDA) for cabotegravir, with the target approval date of January 24, 2022.
Cabotegravir is a long-acting injectable for pre-exposure prophylaxis, or PrEP medication for persons who are at risk of HIV infection and who have had a HIV negative result before starting the drug.
During trials it was found that cabotegravir administered as an intramuscular injection every eight weeks worked better than daily PrEP (oral emtricitabine/tenofovir disoproxil fumarate) pills at preventing HIV. At present, PrEP is taken as a pill daily, on-demand or periodically.
Significant Impact On The Lives Of Gay Men And Trans Women
Shannon Hader, the Deputy Executive Director of the Joint United Nations Program on HIV/AIDS, described the findings as “breakthrough that will have a significant impact on the lives of gay men and other men who have sex with men and transgender women when they are at higher risk of HIV infection.”
The review period for a drug is usually 10 months, but the priority review means this will be brought down to six months. If approved, it will be the first long-acting HIV prevention drug.
“The FDA’s Priority Review designation of cabotegravir long-acting for PrEP underscores the importance of this medicine, supported by the results of the HPTN studies, which demonstrated cabotegravir’s superior efficacy over daily oral FTC/TDF tablets,” Dr Kimberly Smith, , Head of Research & Development at ViiV Healthcare, said in a statement.
Less than 25% of persons in the US who could benefit from PrEP are currently taking it, and ViiV Healthcare said this underscores the need for additional options to prevent HIV.
“We believe new options like investigational cabotegravir long-acting for PrEP will help play a significant role in our collective efforts to end the HIV epidemic,” added Kimberly Smith.
Superior To Daily PrEP Pills
According to ViiV Healthcare, the NDA was based on the results from two phase IIb/III studies, which looked at the safety and efficacy of the medication among 4,566 men who have sex with men and transgender women and 3,223 cisgender women. After an initial oral lead-in phase, cabotegravir was administered as an intramuscular injection every eight weeks.
The independent Data Safety Monitoring Boards stopped the blinded, randomised part of both studies after “cabotegravir was shown to be superior to daily oral emtricitabine/tenofovir disoproxil fumarate (FTC/TDF) tablets in preventing the acquisition of HIV.”
The most common adverse reactions that was observed in at least 1% of the participants included, injection site reactions, diarrhoea, headache, pyrexia, fatigue, sleep disorders, nausea, dizziness, flatulence, and abdominal pain
ViiV Healthcare has not disclosed the pricing details of the new drug.











I have a question is there HIV injection? And if Yes has it began to be used.