
Gay app Hornet backs VAC petition to have PrEP approved in Australia

US-based gay dating/hookup app Hornet has publicly backed Victorian AIDS Council’s (VAC) online petition to have PrEP approved in Australia by promoting it to its users.
The app sent a private message to its users asking for their support, accompanied with a link to the petition that is addressed to federal Health Minister Sussan Ley.
The message read:
“Our friends from down under need our help! Do you believe Truvada for PrEP should be approved for use in other countries outside of the U.S.? If yes, help the Victorian AIDS Council and sign this petition to get it approved in Australia. #approvePrEPdownunder.”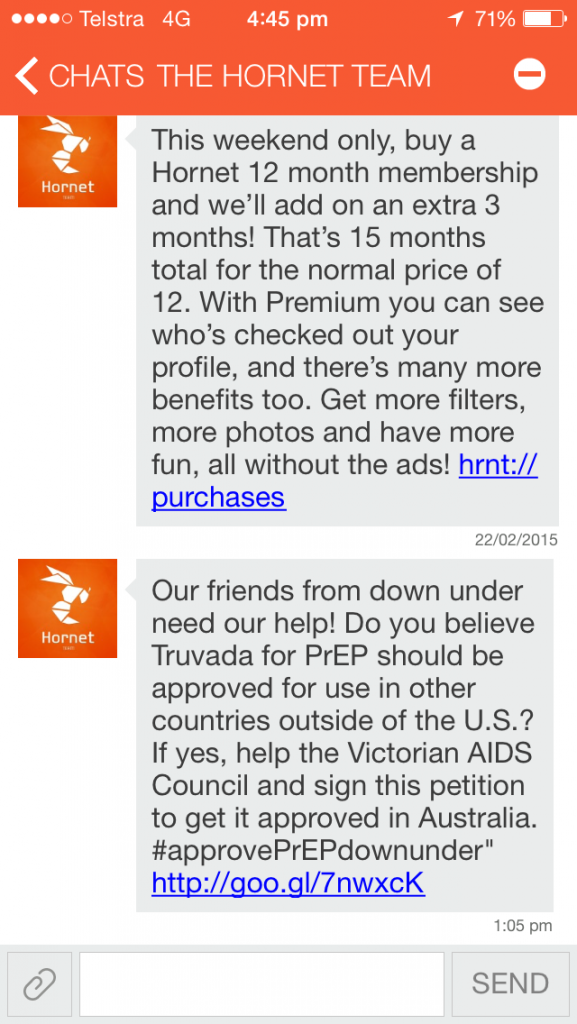
The petition started three days ago and at the time of print, it had amassed 1491 signatures out of the 1500 goal.
Earlier this week, pharmaceutical company Gilead confirmed it would submit an application to the Therapeutic Goods Association (TGA) to have the Truvada drug approved for use as a HIV-prevention method in Australia.
Truvada was approved by the US Food and Drug Administration (FDA) in 2012, and has seen an increasing number of people in the country using it as PrEP (pre-exposure prophylaxis).
The Australian Government recently acknowledged PrEP as a strategy for HIV prevention in the 7th National HIV Strategy 2014-2017.
However, PrEP would not be available at an affordable price should the TGA approve it to begin with, as it would still need to go to the Pharmaceutical Benefits Scheme afterwards for approval.
The VAC outlined in its position statement that they “[support] the approval of PrEP for people who, for whatever reason, are at high risk of acquiring HIV”.
They cite that US-conducted research “overwhlemingly confirms that PrEP works” and the vast benefit that its approval would have on at risk communities in Australia.
In the meantime, PrEP may be acquired through alternative means – outlined in an information sheet published by ACON last month.










Grindr are also doing the same!
Well they would know what messages guys talk about when hooking up