HIV medication Triumeq now available through the PBS
AUSTRALIA’S leading HIV organisations have welcomed today’s announcement that HIV antiviral medication Triumeq is now being available through the Pharmaceutical Benefits Scheme (PBS).
Its listing adds it to the range of treatment options readily available through the PBS for people living with HIV in Australia.
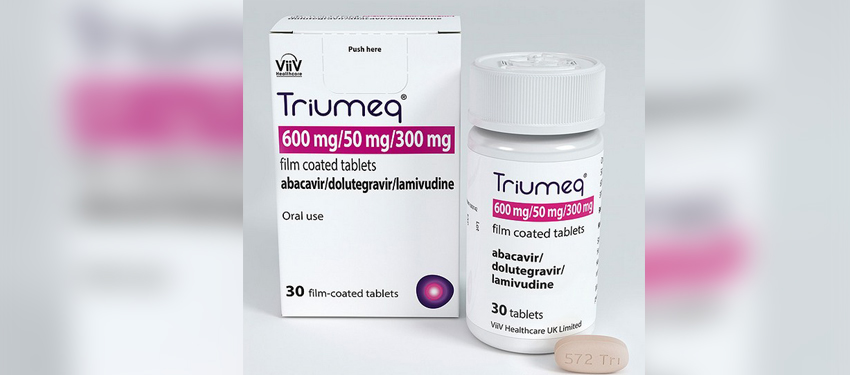
Triumeq is a once-daily tablet combing three antiviral medications: dolutegravir, abacavir and lamivudine, which have been proven to be highly effective in treating HIV. Triumeq is also the first single-pill therapy to become available that contains dolutegravir.
The medication is available to treat HIV in adults and adolescents over 12 years of age.
Aaron Cogle from the National Association of People with HIV Australia, said that HIV treatment guidelines recommend all people with HIV consider taking treatment, both for the individual health benefits and because being on treatment significantly reduces the risk of transmitting the virus.
“To complement these recommendations it is very important that HIV medications are easy to take, potent and have minimal side effects so that people with HIV are able to incorporate HIV treatment into their day to day living with minimal impact,” Cogle said.
“Triumeq provides another choice for doctors and people with HIV to consider in their HIV treatment planning.”
Cogle added that even though there have been advances in the potency, tolerability and convenience of HIV treatments, not all people living with HIV were aware of these.
“Misunderstandings and misconceptions can be a significant barrier for some people with HIV from receiving the benefits of modern HIV treatments, including the belief that HIV treatment can be deferred for long periods and that side-effects and other difficulties are common,” he said.
“Talking these issues through with a doctor experienced in HIV medicine is very important. Local people with HIV organisations can also provide information and there are many excellent resources available online to help people with their treatment decision making.”
Cogle recommended Living Positive Victoria’s Top 10 HIV Treatment Myths and Misconceptions as one of the resources available.
“This resource is designed to assist people with HIV with their treatment decisions, by addressing some of the common misconceptions about taking HIV treatment,” he said.
“We need to ensure that all Australians have access to the latest information on HIV treatment. People with HIV who aren’t already on treatment or who have concerns should start a conversation with their doctor and consider seeking support from their local [HIV organisations].”
Meanwhile, Victorian AIDS Council’s health promotion, policy and communications director Colin Batrouney highlighted the significance of Triumeq’s PBS listing.
“Every advance in HIV medication delivery helps people living with HIV to adhere to their treatment regimens easier,” he said.
“The approval of Triumeq will mean that people will be able to take three drugs in one simple pill.
“Although combination antiretrovirals are nothing new, this is the first time that these three powerful drugs have been put together to fight HIV.”
Kirby Institute director David Cooper said Triumeq could play a key role in improving treatment rates in line with the targets from the National HIV Strategy.
ACON chief executive Nicolas Parkhill echoed the sentiments, saying it complimented the organisation’s [TREAT EARLY] campaign that was launched in February to highlight the benefits of early treatment for HIV – not only for individual health benefit, but also to assist with reducing viral load and therefore HIV transmission risk.
“ACON warmly welcomes the Federal Government’s decision to include Triumeq on the PBS as it will contribute to more accessible HIV treatment,” he said.
“However, there are a range of other important initiatives the Federal Government can support to realise the potential of the latest research evidence, and substantially reduce new HIV infections.
“ACON will particularly continue to support policy and programmatic change in support of more widely available testing options, as well as prevention options such as PrEP.”